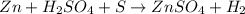Chemistry, 24.06.2021 15:50 chancecharles9oug353
In the following reaction, Zn is Zn(s) H2SO4(aq) --> ZnSO4(aq) H2(g) A. Reduced B. Oxidized C. This is not a redox reaction D. An oxidizing agent

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:40
Determine the energy released per kilogram of fuel used. given mev per reaction, calculate energy in joules per kilogram of reactants. consider 1 mole of tritium plus 1 mole of deuterium to be a mole of “reactions” (total molar mass = 5 grams).
Answers: 1

Chemistry, 22.06.2019 00:30
What is the most stable monatomic ion formed from nitrogen
Answers: 2

Chemistry, 22.06.2019 15:50
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate
Answers: 1

Chemistry, 23.06.2019 00:00
(04.05 hc) analyze the given diagram of the carbon cycle below. part 1: which compound does c represent? part 2: name a process that could release this compound into the air. part 3: explain how the elements that form it are conserved during the carbon cycle. use complete sentences to explain your answer. justify how this compound was created from a recycling of carbon in the carbon cycle. use complete sentences to explain your answer.
Answers: 3
You know the right answer?
In the following reaction, Zn is Zn(s) H2SO4(aq) --> ZnSO4(aq) H2(g) A. Reduced B. Oxidized C. Th...
Questions

Chemistry, 18.10.2020 02:01

Mathematics, 18.10.2020 02:01




Mathematics, 18.10.2020 02:01

Mathematics, 18.10.2020 02:01


Mathematics, 18.10.2020 02:01

Mathematics, 18.10.2020 02:01


English, 18.10.2020 02:01



Mathematics, 18.10.2020 02:01

Engineering, 18.10.2020 02:01

Mathematics, 18.10.2020 02:01



Mathematics, 18.10.2020 03:01