
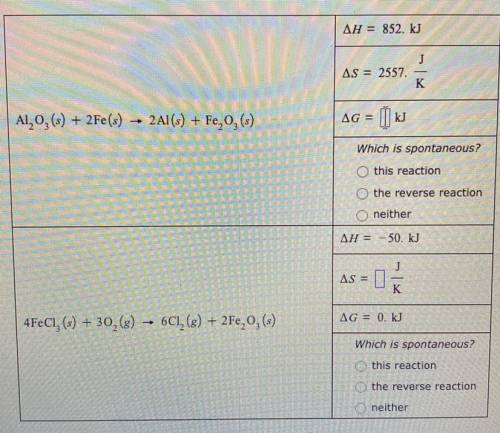
A chemical engineer is studying the two reactions shown in the table below.
In each case, she fills a reaction vessel with some mixture of the reactants and products at a constant temperature of 57.0°C and constant total pressure. Then, she measures that reaction enthalpy delta H and the reaction entropy delta S of the first reaction, and the reaction enthalpy delta H and reaction free energy delta G of the second reaction. The results of her measurements are shown in the table.
Complete the table. That is, calculate delta G for the first reaction and delta S for the second. (Round your answer to zero decimal places.) Then, decide whether, under the conditions the engineer has set up, the reaction is spontaneous, the reverse reaction is spontaneous, or neither forward or reverse reaction is spontaneous because the system is at equilibrium.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:00
The tilt of the earth's axis of rotation is responsible for the a) ocean's tides. b) size of the moon. c) brightness of stars. d) earth’s seasons.
Answers: 1

Chemistry, 23.06.2019 02:30
Which words or phrases identify layers of groundwater? check all that apply. water table kettle lake saturation zone underground lake sinkhole will give brainiest, answer quickly.
Answers: 1

Chemistry, 23.06.2019 04:00
What changes occur in the reaction indicated by the equation? check all that apply. the hydrogen nucleus loses protons. the oxygen nucleus gains protons. the bond in h2 is broken, and new bonds are formed between hydrogen and oxygen atoms. each electron associated with a hydrogen atom is shared with an oxygen atom.
Answers: 3

Chemistry, 23.06.2019 06:30
How can the number of core electrons be determined from the periodic table
Answers: 1
You know the right answer?
A chemical engineer is studying the two reactions shown in the table below.
In each case, she fills...
Questions


Mathematics, 20.10.2020 21:01

Mathematics, 20.10.2020 21:01

Biology, 20.10.2020 21:01



Biology, 20.10.2020 21:01


English, 20.10.2020 21:01



Social Studies, 20.10.2020 21:01

Mathematics, 20.10.2020 21:01

Mathematics, 20.10.2020 21:01


Chemistry, 20.10.2020 21:01



English, 20.10.2020 21:01






