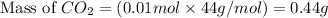Chemistry, 29.06.2021 02:10 fjjjjczar8890
Calculate the number of molecules of carbon (IV) oxide
produced when 10g of CaCO3 is treated with 100cm of
0.2mol dm HCl. The equation of the reaction is
CaCO3(s) + 2HCl(aq) → CaCl2(aq) + H2O(1) + CO2(g)
ICa=40 (=12 O=161

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

Chemistry, 22.06.2019 12:50
What is the chemical name of the compound na2co3? use the list of polyatomic ions and the periodic table to you answer. a. sodium carbon oxide b. sodium carbonate c. sodium(ll) carbonate d. sodium oxalate
Answers: 1
You know the right answer?
Calculate the number of molecules of carbon (IV) oxide
produced when 10g of CaCO3 is treated with 1...
Questions




Mathematics, 24.04.2021 17:00



History, 24.04.2021 17:00

Mathematics, 24.04.2021 17:00

Arts, 24.04.2021 17:00




Computers and Technology, 24.04.2021 17:10

Biology, 24.04.2021 17:10

Mathematics, 24.04.2021 17:10


Biology, 24.04.2021 17:10

Social Studies, 24.04.2021 17:10


History, 24.04.2021 17:10

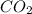 produced is 0.44 g
produced is 0.44 g ......(1)
......(1)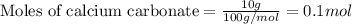
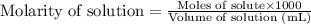 .....(2)
.....(2)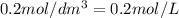 (Conversion factor:
(Conversion factor: 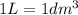
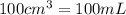 (Conversion factor:
(Conversion factor: 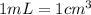
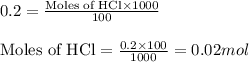
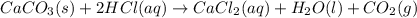
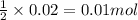 of calcium carbonate
of calcium carbonate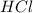 produces 1 mole of
produces 1 mole of