
Chemistry, 30.06.2021 02:10 alesiabarrios6
Na3N decomposes to form sodium and nitrogen gas at STP. If 13.7 L of nitrogen is produced
how many moles of Na3N was used? (22.4 L = 1 mole of any gas)
2Na3N --> 6Na + N2

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:40
For a patient with the following pes statement and interventions, which would be the most appropriate monitoring and evaluating data? pes statement: inadequate calcium intake related to food and nutrition related knowledge deficit as evidenced by statements that the only dietary source of calcium is milk and she believes that she is lactose intolerant. patient’s nutrition prescription is for a diet providing 1200 mg calcium per day. patient was provided with in-depth nutrition education on alternative dietary and supplement sources of calcium. a. calcium intake (at subsequent visit) b. knowledge assessment by asking patient to identify food sources from menus and shopping list (at the end of the current visit) c. serum calcium (at next visit) d. both a and b e. both a and c
Answers: 2

Chemistry, 22.06.2019 08:30
The mass of a neutron is equal to the mass of a proton plus the mass of an electron. true or false false true
Answers: 1

Chemistry, 22.06.2019 21:00
Kp is the equilibrium constant for dissociation of the propionic acid dimer. what is the sign of the slope for a plot of the natural logarithm of kp vs. inverse temperature for this reaction?
Answers: 1

Chemistry, 23.06.2019 04:10
Two solids are mixed in a flask and stirred. after a few minutes, the flask becomes cold. which of the following best describes this reaction? a. an exothermic reaction b. a combustion reaction c. an endothermic reaction d. a decomposition reaction
Answers: 1
You know the right answer?
Na3N decomposes to form sodium and nitrogen gas at STP. If 13.7 L of nitrogen is produced
how many...
Questions



History, 08.04.2020 01:16

Biology, 08.04.2020 01:16



Arts, 08.04.2020 01:16

Biology, 08.04.2020 01:16


English, 08.04.2020 01:16


Mathematics, 08.04.2020 01:16



Mathematics, 08.04.2020 01:16




Mathematics, 08.04.2020 01:16

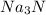 were used.
were used.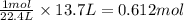
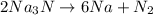
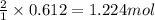 of
of 

