Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Fission of uranium-235 products energy and a. isotopes of smaller elements b. isotopes of larger elements c. lighter isotopes of uranium d. heavier isotopes of uranium
Answers: 3

Chemistry, 22.06.2019 05:00
Given sno2 + 2h2 - sn + 2h20 tin oxide reacts with hydrogen to produce tin and water. how many moles of sno2 are needed to produce 500.0 grams of sn?
Answers: 3

Chemistry, 22.06.2019 09:40
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3

Chemistry, 22.06.2019 22:30
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3
You know the right answer?
Calculate the vapor pressure (in torr) at 298 K in a solution prepared by dissolving 46.8 g of the n...
Questions






History, 08.12.2021 22:10


Mathematics, 08.12.2021 22:10

Chemistry, 08.12.2021 22:10

Biology, 08.12.2021 22:10

Health, 08.12.2021 22:10

Social Studies, 08.12.2021 22:10

English, 08.12.2021 22:10

Social Studies, 08.12.2021 22:10

Business, 08.12.2021 22:10

Social Studies, 08.12.2021 22:10

History, 08.12.2021 22:10

English, 08.12.2021 22:10

Mathematics, 08.12.2021 22:10

Spanish, 08.12.2021 22:10

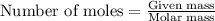 ......(1)
......(1)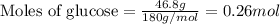
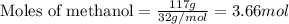
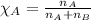 .....(2)
.....(2)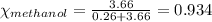
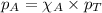 .....(3)
.....(3)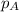 is the partial pressure of component A in the mixture and
is the partial pressure of component A in the mixture and 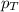 is the total partial pressure of the mixture
is the total partial pressure of the mixture