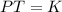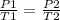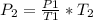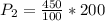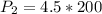Chemistry, 02.07.2021 01:00 1tallison1
A gas has a pressure of 450 mmHg at 100 degrees Celsius. What will its new pressure be when the temperature rises 200 degrees Celsius

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:30
Which compound contains both ionic and covalent bonds? a) hbr b)cbr4 c)nabr d) naoh
Answers: 2

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 13:30
How many protons, electrons, and neutrons are in each of the following isotopes? a. zirconium-90 b. palladium-108 c. bromine-81 d. antimony-123
Answers: 1
You know the right answer?
A gas has a pressure of 450 mmHg at 100 degrees Celsius. What will its new pressure be when the temp...
Questions



Mathematics, 11.11.2021 14:00




Mathematics, 11.11.2021 14:00

Mathematics, 11.11.2021 14:00



Physics, 11.11.2021 14:00


Social Studies, 11.11.2021 14:00



History, 11.11.2021 14:00

Social Studies, 11.11.2021 14:00

Social Studies, 11.11.2021 14:00

Mathematics, 11.11.2021 14:00