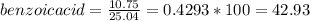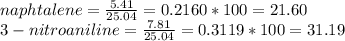Chemistry, 02.07.2021 04:40 lydiapoetz5330
1. Calculate the percent recovery of benzoic acid, naphthalene and 3-nitroaniline if you were able to collect 10.75 g of benzoic acid, 5.41 g of naphthalene, and 7.81 g of 3-nitroaniline from a set of extractions. The starting mass of the mixture was 25.04 g. (0.6 pt) 2. Describe why it is important to use sodium hydroxide and hydrochloride acid in this experiment. Why was it necessary to initially start off with a 5% solution of the acid or base for this experiment

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 16:30
An atom with 7 protons, 6 neutrons, and 7 electrons has an atomic mass of amu. (enter a whole number.) numerical answers expected! answer for blank 1:
Answers: 3

Chemistry, 23.06.2019 00:30
Element j is 1s 2s 2p 3s . (i) how many unpaired electrons does j have? (ii) is j a good oxidizing agent or a reducing agent? (iii) state reason for the answer.
Answers: 1

Chemistry, 23.06.2019 04:20
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1
You know the right answer?
1. Calculate the percent recovery of benzoic acid, naphthalene and 3-nitroaniline if you were able t...
Questions

Mathematics, 29.06.2019 16:30


Mathematics, 29.06.2019 16:30









History, 29.06.2019 16:30