Answers: 2


Another question on Chemistry

Chemistry, 23.06.2019 04:00
Achemical reaction is done in the setup shown , resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 2

Chemistry, 23.06.2019 14:00
Fill in the blanks for the following statements: the rms speed of the molecules in a sample of h2 gas at 300 k will be times larger than the rms speed of o2 molecules at the same temperature, and the ratio µrms (h2) / µrms (o2) with increasing temperature.
Answers: 1

Chemistry, 23.06.2019 17:00
What statement about the energy of a phase change is true
Answers: 1

Chemistry, 23.06.2019 22:00
2. a sample of table sugar (sucrose, c12h22o11) has a mass of 1.202 g. a. calculate the number of moles of c12h22o11 contained in the sample. show your work. b. calculate the moles of each element in c12h22o11, show your work c. calculate the number of atoms of each type in c12h22o11. show your work
Answers: 2
You know the right answer?
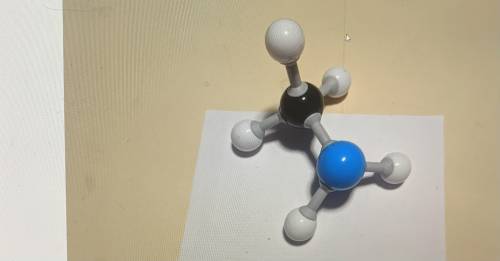
What molecule is this
...
...
Questions

Physics, 28.10.2019 02:31


Mathematics, 28.10.2019 02:31


Mathematics, 28.10.2019 02:31


Mathematics, 28.10.2019 02:31

Mathematics, 28.10.2019 02:31

Mathematics, 28.10.2019 02:31



Mathematics, 28.10.2019 02:31

Mathematics, 28.10.2019 02:31




Mathematics, 28.10.2019 02:31


Mathematics, 28.10.2019 02:31