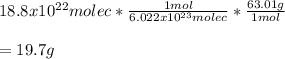Chemistry, 08.07.2021 04:40 reneebrown017
A sample of hydrogen nitrate or nitric acid, HNO 3 contains 18.8 x 1022 molecules. How much mass of nitric acid are in the sample?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 22:30
Which of the following molecules is polar? c3h7oh c2h5cooh
Answers: 1

Chemistry, 23.06.2019 00:30
Ok, so i have 2 questions. try to answer them both: (the topic is fire) 1) how can you represent the chemical reaction of fire? 2) what kind of bond is formed in this chemical reaction
Answers: 3

Chemistry, 23.06.2019 01:30
Magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. trial 1: trial 2: data mass of empty crucible with lid trial 1: 26.688 trial 2: 26.681 mass of mg metal, crucible, and lid trial 1: 26.994 trial: 2 26.985 mass of mgo, crucible, and lid trial 1: 27.188 trial 2: 27.180
Answers: 1
You know the right answer?
A sample of hydrogen nitrate or nitric acid, HNO 3 contains 18.8 x 1022 molecules.
How much mass of...
Questions


English, 25.08.2019 07:30

Mathematics, 25.08.2019 07:30

Social Studies, 25.08.2019 07:30

Mathematics, 25.08.2019 07:30


Mathematics, 25.08.2019 07:30


History, 25.08.2019 07:30

History, 25.08.2019 07:30


English, 25.08.2019 07:30

Biology, 25.08.2019 07:30

Physics, 25.08.2019 07:30


World Languages, 25.08.2019 07:30

Biology, 25.08.2019 07:30

Chemistry, 25.08.2019 07:30