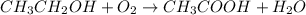Wine goes bad soon after opening because the ethanol dissolved in it reacts with oxygen gas to form water and aqueous acetic acid , the main ingredient in vinegar. Calculate the moles of acetic acid produced by the reaction of of ethanol. Be sure your answer has a unit symbol, if necessary, and round it to significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:00
__ _ _ _ _ is the process of removing earth materials from their original sites through weathering and transport and depositing the in another location. a. erosion b. sedimentation c. lithification d. dissolution
Answers: 1


Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3

Chemistry, 22.06.2019 19:20
The equation picture below shows which type of nuclear reaction u 235 + n x e 134 + sr 100 + 2n
Answers: 1
You know the right answer?
Wine goes bad soon after opening because the ethanol dissolved in it reacts with oxygen gas to form...
Questions





Mathematics, 20.03.2020 03:10

Mathematics, 20.03.2020 03:10

Mathematics, 20.03.2020 03:10






English, 20.03.2020 03:11



Mathematics, 20.03.2020 03:11


History, 20.03.2020 03:11


Chemistry, 20.03.2020 03:11