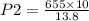Chemistry, 09.07.2021 16:30 annrawcliffe
A 10.0 L balloon contains helium gas at a pressure of 655 mmHg. What is the final pressure, in millimeters of mercury, if the final volume is 13.8 L? Please show your work in order to receive credit.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:30
You have 125g of a certain seasoning and are told that it contains 76.0 g of salt what is the percentage of salt by mass in this seasoning
Answers: 1

Chemistry, 22.06.2019 03:30
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1

Chemistry, 22.06.2019 05:50
What are the 4 phases of matter in order of increasing engery content?
Answers: 2

Chemistry, 22.06.2019 05:50
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1
You know the right answer?
A 10.0 L balloon contains helium gas at a pressure of 655 mmHg. What is the final pressure, in milli...
Questions

Mathematics, 30.03.2021 20:50

Mathematics, 30.03.2021 20:50


Mathematics, 30.03.2021 20:50

Mathematics, 30.03.2021 20:50

Chemistry, 30.03.2021 20:50

Mathematics, 30.03.2021 20:50

Mathematics, 30.03.2021 20:50


Business, 30.03.2021 20:50





Advanced Placement (AP), 30.03.2021 20:50

Mathematics, 30.03.2021 20:50

Mathematics, 30.03.2021 20:50


Mathematics, 30.03.2021 20:50