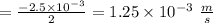Chemistry, 11.07.2021 18:50 sammuelanderson1371
Consider the reaction “2 SO2 (g) + O2 (g) = 2 SO3 was 0.175 M. After 50 s the concentration of SO2 Date: (g)”. Initial concentration of SO2 (g) (g) became 0.0500 M. Calculate rate of the reaction

Answers: 2


Another question on Chemistry

Chemistry, 23.06.2019 00:00
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1


Chemistry, 23.06.2019 07:10
Which one of the following is an oxidation-reduction reaction? naoh + hno3 --> h2o + kno3 naoh + hno3 --> h2o + kno3 so3 + h2o --> h2so4 cacl2 + na2co3 --> caco3 + 2 nacl ch4 + 2 o2 --> co2 + 2 h2o al2(so4)3 + 6 koh --> 2 al(oh)3 + 3 k2so4
Answers: 3

Chemistry, 23.06.2019 11:30
How do you calculate the mass of a product when the amounts of more than one reactant are given?
Answers: 3
You know the right answer?
Consider the reaction “2 SO2 (g) + O2 (g) = 2 SO3 was 0.175 M. After 50 s the concentration of SO2 D...
Questions

English, 18.03.2022 20:30



Physics, 18.03.2022 20:40

Mathematics, 18.03.2022 20:40

Physics, 18.03.2022 20:40



Geography, 18.03.2022 20:40


Spanish, 18.03.2022 20:40




Chemistry, 18.03.2022 20:40

Mathematics, 18.03.2022 20:50



Mathematics, 18.03.2022 20:50

 "
"![=-\frac{1}{2} \frac{\Delta [SO_2]}{\Delta t} =-\frac{\Delta [O_2]}{\Delta t}= +\frac{1}{2} \frac{\Delta [SO_3]}{\Delta t}\\\\=\frac{\Delta [SO_2]}{\Delta t}=\frac{0.0500-0.175\ M}{505}= -2.5 \times 10^{-3} \ \frac{m}{s}\\\\](/tpl/images/1392/3786/a8505.png)