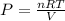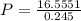Chemistry, 11.07.2021 19:10 Division101
A 0.245-L flask contains 0.467 mol co2 at 159 °c. Calculate the pressure using the ideal gas law.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. the values of phosphorous acid are 1.30 6.70 calculate the ph for each of the given points in the titration of 50.0 ml of 1.5 m h3po3(aq) with 1.5 m koh(aq) .
Answers: 3

Chemistry, 22.06.2019 05:30
The climate of the continental united states is generally 1. tropical 2. temperate 3. arctic 4. highland
Answers: 1

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 22.06.2019 20:00
What is the molarity of the solution produced when 145 g of nacl is dissolved in sufficient water to prepare 2.75 l of solution?
Answers: 1
You know the right answer?
A 0.245-L flask contains 0.467 mol co2 at 159 °c. Calculate the pressure using the ideal gas law....
Questions

History, 20.07.2019 01:30

Social Studies, 20.07.2019 01:30

History, 20.07.2019 01:30

Physics, 20.07.2019 01:30

Chemistry, 20.07.2019 01:30

Mathematics, 20.07.2019 01:30



Social Studies, 20.07.2019 01:30

English, 20.07.2019 01:30

Health, 20.07.2019 01:30


History, 20.07.2019 01:30


Mathematics, 20.07.2019 01:30

Biology, 20.07.2019 01:30

English, 20.07.2019 01:30


History, 20.07.2019 01:30