
In aqueous solution the Hg2+ ion forms a complex with four iodide anions. Write the formation constant expression for the equilibrium between the hydrated metal ion and the aqueous complex. Under that, write the balanced chemical equation for the first step in the formation of the complex.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:10
Which of the following elements would you expect to have the lowest ionization energy value? fluorine, lithium, neon, nitrogen
Answers: 2

Chemistry, 22.06.2019 18:30
The number of moles of a given mass of a substance can be found without knowing its molecular formula or molar mass. true false
Answers: 1

Chemistry, 22.06.2019 21:20
If a simple machine aduces the strength of a force, what must be increased? the speed of the input force the work the simple machine performs the size of the simple machine the distance over which the force is applied
Answers: 1

Chemistry, 23.06.2019 03:30
Why do electrons further from the nucleus have more energy
Answers: 1
You know the right answer?
In aqueous solution the Hg2+ ion forms a complex with four iodide anions. Write the formation consta...
Questions




Mathematics, 28.06.2019 01:00



Mathematics, 28.06.2019 01:00


Mathematics, 28.06.2019 01:00

History, 28.06.2019 01:00



English, 28.06.2019 01:00

Mathematics, 28.06.2019 01:00

Mathematics, 28.06.2019 01:00


Mathematics, 28.06.2019 01:00



Mathematics, 28.06.2019 01:00

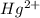 ion is used to represent a
ion is used to represent a ![[Hg(H_2O)_4]^{2+}](/tpl/images/1394/5126/65dfb.png) .
.
![[Hgl_4]^{2-}](/tpl/images/1394/5126/970b6.png) .
. ![[Hg(H_2O)_4]^{2+} +4I^{-} \to [HgI_4]^{2-}+4H_20](/tpl/images/1394/5126/c93e6.png)
![k_f=\frac{[HgI_4]^{2-}}{[Hg(H_2O)_4]^{2+} [I^{-}]^4}](/tpl/images/1394/5126/9ffc5.png)
![[Hg(H_2O)_4]^{2+} +I^{-} \to [HgI(H_2O)_3]^{+}+H_20](/tpl/images/1394/5126/c074d.png)


