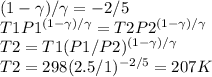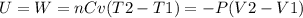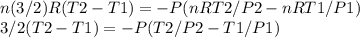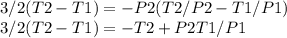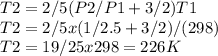Chemistry, 15.07.2021 18:40 zoeheskett
A quantity of 0.27 mole of neon is confined in a container at 2.50 atm and 298 Kand then allowed to expand adiabatically under two different conditions: (a) reversibly to 1.00 atm and (b) against a constant pressure of 1.00 atm. Calculate the final temperature in each case.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:30
What is the force of attraction between the particles in a salt crystal
Answers: 2

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 09:30
Why do cells appear different in distilled water than they do in 10% salt water?
Answers: 2

Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1
You know the right answer?
A quantity of 0.27 mole of neon is confined in a container at 2.50 atm and 298 Kand then allowed to...
Questions

French, 17.09.2019 08:30

Mathematics, 17.09.2019 08:30


Computers and Technology, 17.09.2019 08:30


Mathematics, 17.09.2019 08:30



Mathematics, 17.09.2019 08:30

Mathematics, 17.09.2019 08:30



Chemistry, 17.09.2019 08:30

Biology, 17.09.2019 08:30

Mathematics, 17.09.2019 08:30

Chemistry, 17.09.2019 08:30

Mathematics, 17.09.2019 08:30