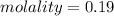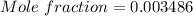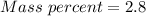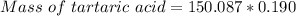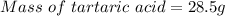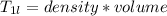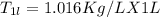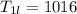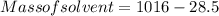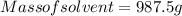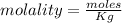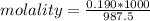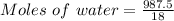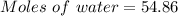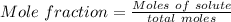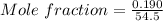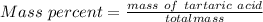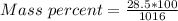Chemistry, 16.07.2021 01:00 911wgarcia
A beverage contains tartaric acid, H2C4H4O6, a substance obtained from grapes during wine making. If the beverage is 0.190 tartaric acid, what is the molal concentration? What is the mole fraction of tartaric acid and water? Calculate the mass percent of tartaric acid. The density of the solution is 1.016g/mL.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3

Chemistry, 22.06.2019 15:30
The identities of substances are the same before and after which type of change
Answers: 1

Chemistry, 22.06.2019 18:00
How does climate change cause the ocean's thermohaline current to slow down?
Answers: 3

Chemistry, 22.06.2019 20:00
If one fission reaction of a uranium-235 atom produced two neutrons, how many neutrons would be released if the chain reaction occurred three more times?
Answers: 1
You know the right answer?
A beverage contains tartaric acid, H2C4H4O6, a substance obtained from grapes during wine making. If...
Questions

Mathematics, 05.01.2022 23:30






Engineering, 05.01.2022 23:30

Mathematics, 05.01.2022 23:30

Mathematics, 05.01.2022 23:30




Chemistry, 05.01.2022 23:40

Mathematics, 05.01.2022 23:40



History, 05.01.2022 23:40



Mathematics, 05.01.2022 23:40