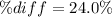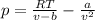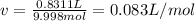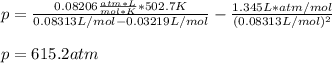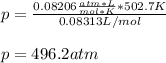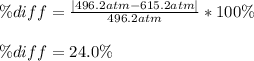Chemistry, 18.07.2021 03:50 andrea1704
According to the ideal gas law, a 9.998 mol sample of argon gas in a 0.8311 L container at 502.7 K should exert a pressure of 496.2
atm. What is the percent difference between the pressure calculated using the van der Waals' equation and the ideal pressure? For Ar
gas, a = 1.345 L’atm/mol? and b = 3.219x10-2 L/mol.
Pideal – Puan der Waals |
Percent difference
x 100

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 16:20
When water dissolves sugar, which process is not involved? o dissociation o hydration o surface area of the solute increases sa
Answers: 1

Chemistry, 22.06.2019 16:30
Explain in detail of the four major scientific developments that spurred the formulation of the plate tectonics theory
Answers: 2

Chemistry, 22.06.2019 20:00
State one important difference between a physical change and a chemical change?
Answers: 1
You know the right answer?
According to the ideal gas law, a 9.998 mol sample of argon gas in a 0.8311 L container at 502.7 K s...
Questions


Mathematics, 23.03.2021 01:10



History, 23.03.2021 01:10



Mathematics, 23.03.2021 01:10





English, 23.03.2021 01:10

Mathematics, 23.03.2021 01:10


Spanish, 23.03.2021 01:10