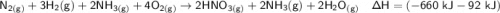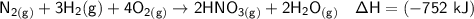Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. In the first step, nitrogen and hydrogen react to form ammonia: N2 (g) + 3H2 (g) â 2NH3 (g) =ÎHâ92.kJ In the second step, ammonia and oxygen react to form nitric acid and water:
NH3 (g) + 2O2 (g) â HNO3 (g) + H2O (g) =ÎHâ330.kJ
Required:
Calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 01:30
(apex) when a cup of water is dropped, as the cup falls, the water in the cup falls out true or false?
Answers: 1

Chemistry, 22.06.2019 05:50
What are transitions between a liquid and a solid called? identify which way they are transitioning
Answers: 2

Chemistry, 22.06.2019 06:30
Design techniques and materials that reduce the negative environmental impact of a structure are referred to as
Answers: 2
You know the right answer?
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen...
Questions



Biology, 31.07.2020 14:01



Mathematics, 31.07.2020 14:01

Mathematics, 31.07.2020 14:01


Computers and Technology, 31.07.2020 14:01



Mathematics, 31.07.2020 14:01



World Languages, 31.07.2020 14:01



Mathematics, 31.07.2020 14:01

Mathematics, 31.07.2020 14:01

Mathematics, 31.07.2020 14:01

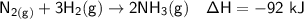 ---- (1)
---- (1)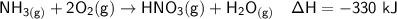 ---- (2)
---- (2)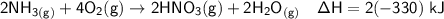 ----- (4)
----- (4)