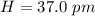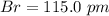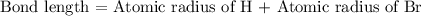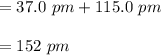Chemistry, 19.07.2021 18:00 nakeakohler13
Bond length is the distance between the centers of two bonded atoms. On the potential energy curve, the bond length is the internuclear distance between the two atoms when the potential energy of the system reaches its lowest value. Given that the atomic radii of H and Br are 37.0 pm and 115 pm , respectively, predict the upper limit of the bond length of the HBr molecule. Express your answer to three significant figures and include the appropriate units. View Available Hint(s)for Part C

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3

Chemistry, 22.06.2019 17:00
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 3

Chemistry, 22.06.2019 21:00
In the experiment you asked to react hydrochloric acid and with sodium hydroxide. when measuring the volume of the reactants, which instrument would give the greatest precision.
Answers: 3
You know the right answer?
Bond length is the distance between the centers of two bonded atoms. On the potential energy curve,...
Questions

Spanish, 23.09.2020 06:01





History, 23.09.2020 06:01

Mathematics, 23.09.2020 06:01

English, 23.09.2020 06:01



Mathematics, 23.09.2020 06:01





Mathematics, 23.09.2020 06:01

Biology, 23.09.2020 06:01


History, 23.09.2020 06:01

Mathematics, 23.09.2020 06:01