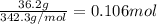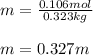Chemistry, 19.07.2021 19:10 nhester2056
Calculate the molality of each of the following solutions: (a) 36.2 g of sucrose (C12H22O11) in 323 g of water, m (b) 8.63 moles of ethylene glycol (C2H6O2) in 1889 g of water.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1

Chemistry, 23.06.2019 03:20
High-pressure liquid chromatography (hplc) is a method used in chemistry and biochemistry to purify chemical substances. the pressures used in this procedure range from around 500 kilopascals (500,000 pa) to about 60,000 kpa (60,000,000 pa). it is often convenient to know the pressure in torr. if an hplc procedure is running at a pressure of 1.03×108 pa , what is its running pressure in torr?
Answers: 3

Chemistry, 23.06.2019 04:10
What does the field of thermodynamics relate to a-changes in nuclear reactions b- changes in energy in systems c changes in molecular structure d changes in atomic properties
Answers: 1

Chemistry, 23.06.2019 05:00
Select the statement that describe chemical properties a. antacid tablets neutralize stomach acid b. helium is the lightest monatomic element c. water freezes at 0 celsius d. mercury is liquid at room temperature
Answers: 3
You know the right answer?
Calculate the molality of each of the following solutions: (a) 36.2 g of sucrose (C12H22O11) in 323...
Questions

Mathematics, 11.03.2021 02:00


Mathematics, 11.03.2021 02:00



Advanced Placement (AP), 11.03.2021 02:00

Physics, 11.03.2021 02:00

Mathematics, 11.03.2021 02:00



Mathematics, 11.03.2021 02:00



Chemistry, 11.03.2021 02:00


Computers and Technology, 11.03.2021 02:00

Computers and Technology, 11.03.2021 02:00



Mathematics, 11.03.2021 02:00