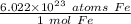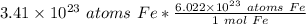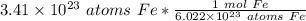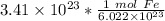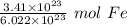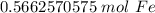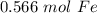Explanation:
We are asked to convert a number of atoms to moles.
We can convert atoms to moles using Avogadro's Number, which is 6.022 × 10²³. This is the number of particles (atoms, molecules, formula units, etc.) in 1 mole of a substance. In this problem, the particles are atoms of iron (Fe). There are 6.022 ×10²³ atoms of iron in 1 mole of iron.
We use dimensional analysis to convert atoms to moles. This involves setting up ratios. Use Avogadro's Number and the underlined information to make a ratio.
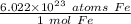
We are converting 3.41 × 10²³ atoms of iron to moles, so we multiply by this value.
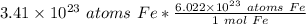
Flip the ratio. It stays equivalent, but it allows the units of atoms of iron to cancel.
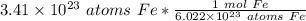
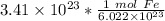
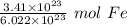
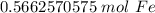
The original measure ment of iron atoms ( 3.41 × 10²³ ) has 3 significant figures, so our answer must have the same. For the number we calculated, that is the thousandths place. The 2 in the ten-thousandths place ( 0.5662570575) tells us to leave the 6 in the thousandths place.
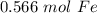
3.41 × 10²³ atoms of iron is equal to approximately 0.566 moles of iron.