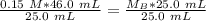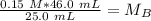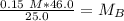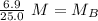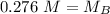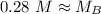Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

Chemistry, 22.06.2019 18:30
The number of moles of a given mass of a substance can be found without knowing its molecular formula or molar mass. true false
Answers: 1


Chemistry, 23.06.2019 06:30
Generally, observed behavior that can be formulated into a statement, sometimes mathematical in nature, is called a(n): a. observation. b. measurement. c. theory. d. natural law. e. experiment.
Answers: 2
You know the right answer?
A titration is performed on a 25.0 mL sample of calcium hydroxide. A volume of
46.0 mL of a 0.15 M...
Questions

English, 06.11.2020 21:30




Social Studies, 06.11.2020 21:30


Arts, 06.11.2020 21:30

History, 06.11.2020 21:30

Mathematics, 06.11.2020 21:30





Social Studies, 06.11.2020 21:30


English, 06.11.2020 21:30



Chemistry, 06.11.2020 21:30


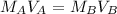

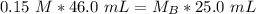
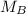 . It is being multiplied by 25.0 milliliters. The inverse operation of multiplication is division, so we divide both sides of the equation by 25.0 mL.
. It is being multiplied by 25.0 milliliters. The inverse operation of multiplication is division, so we divide both sides of the equation by 25.0 mL.