Chemistry, 27.07.2021 22:20 plantkiana677oxa6hk
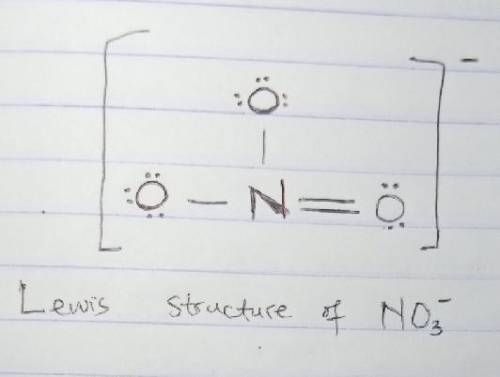
Lewis Structures are used to describe the covalent bonding in molecules and ions. Draw a Lewis structure for NO3- and answer the following questions based on your drawing.
1. For the central nitrogen atom:
The number of lone pairs =
The number of single bonds=
The number of double bonds=
2. The central nitrogen atom :

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
For ai it's atomic number is 13 and it's mass number is 27 how many neutrons does it have
Answers: 1

Chemistry, 22.06.2019 11:30
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2

Chemistry, 22.06.2019 11:30
Compare and contrast refraction of light and sound will give brainliest
Answers: 1

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3
You know the right answer?
Lewis Structures are used to describe the covalent bonding in molecules and ions. Draw a Lewis struc...
Questions


Mathematics, 10.02.2021 19:20

History, 10.02.2021 19:20

Computers and Technology, 10.02.2021 19:20

Mathematics, 10.02.2021 19:20

Mathematics, 10.02.2021 19:20

English, 10.02.2021 19:20

Geography, 10.02.2021 19:20



History, 10.02.2021 19:20


Arts, 10.02.2021 19:20

History, 10.02.2021 19:20


Mathematics, 10.02.2021 19:20



Mathematics, 10.02.2021 19:20

Health, 10.02.2021 19:20