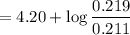Chemistry, 28.07.2021 03:00 nauticatyson9
An analytical chemist is titrating of a solution of benzoic acid with a solution of . The of benzoic acid is . Calculate the pH of the acid solution after the chemist has added of the solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of solution added.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2

Chemistry, 22.06.2019 01:00
The diagram shows the positions of the sun, moon and earth during spring tides, when the high tides are at their highest and low tides at their lowest. what is it about these positions that causes these high and low tides?
Answers: 3

Chemistry, 22.06.2019 06:30
(1.6 × 10-19)(5.0 × 106) = c × 10d identify the missing numbers below to show the result of multiplying the numbers.
Answers: 1

You know the right answer?
An analytical chemist is titrating of a solution of benzoic acid with a solution of . The of benzoic...
Questions

Mathematics, 01.08.2019 17:30

History, 01.08.2019 17:30


Mathematics, 01.08.2019 17:30


Mathematics, 01.08.2019 17:30

Mathematics, 01.08.2019 17:30

Mathematics, 01.08.2019 17:30




History, 01.08.2019 17:30

Computers and Technology, 01.08.2019 17:30

Social Studies, 01.08.2019 17:30


Mathematics, 01.08.2019 17:30

English, 01.08.2019 17:30

Social Studies, 01.08.2019 17:30

Geography, 01.08.2019 17:30

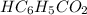 with a 0.3600 M solution of KOH. The
with a 0.3600 M solution of KOH. The 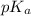 of benzoic acid is 4.20. Calculate the pH of the acid solution after the chemist has added 232.0 mL of the KOH solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of KOH solution added.
of benzoic acid is 4.20. Calculate the pH of the acid solution after the chemist has added 232.0 mL of the KOH solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of KOH solution added.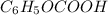
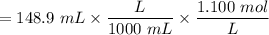
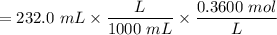
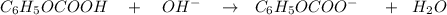
![$C_6H_5OCOOH, [C_6H_5OCOOH]$](/tpl/images/1400/7778/75042.png)
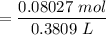
![$C_6H_5OCOO^- , [C_6H_5OCOO^-] =\frac{0.08352 \ mol}{0.3809 \ L}](/tpl/images/1400/7778/c3e04.png)
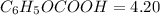
![$pH = pK_a + \log \frac{[C_6H_5OCOO^-]}{[C_6H_5OCOOH]}](/tpl/images/1400/7778/ac810.png)