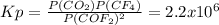Chemistry, 30.07.2021 02:40 blazecarley
Predict whether reactants or products will be favored at equilibrium for the below reaction.
Kp= 2.2 x 10^6 at 298K
2COF2 (g) + ⇌ CO2(g) + CF4(g)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:00
Drag each number to the correct location on the equation. each number can be used more than once, but not all numbers will be used. balance the equation with the coefficients. 2 3 4 5 kclo3 -> kcl + o2
Answers: 1

Chemistry, 21.06.2019 20:20
Concerning the 10.0 ml of 0.50 m nacl to 100 ml of solution: when a solution is diluted, does it change the number of moles dissolved?
Answers: 3

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 15:00
Which of the following is the correct formula for copper (i) sulfate trihydrate? cuso4 · 3h2o cuso4(h2o)3 cu2so4(h2o)3 cu2so4 · 3h2o
Answers: 1
You know the right answer?
Predict whether reactants or products will be favored at equilibrium for the below reaction.
Kp= 2....
Questions

Mathematics, 11.10.2019 18:30





Mathematics, 11.10.2019 18:30


Biology, 11.10.2019 18:30





Biology, 11.10.2019 18:30



Mathematics, 11.10.2019 18:30



Mathematics, 11.10.2019 18:30