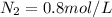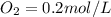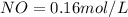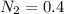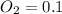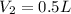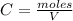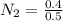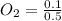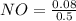Chemistry, 30.07.2021 22:50 Animallover100
A 1 L container originally holds 0.4 mol of N2, 0.1 mol of O2, and 0.08 mole of NO. If the volume of the container holding the equilibrium mixture of N2, O2, and NO is decreased to 0.5 L without changing the quantities of the gases present, how will their concentrations change

Answers: 1


Another question on Chemistry

Chemistry, 23.06.2019 00:30
On the periodic table, elements are arranged by which of the following. a. mass numbers. b. increasing atomic number. c. alphabetical order. or d. density
Answers: 1

Chemistry, 23.06.2019 02:30
Which statement best describes the liquid state of matter? a. it has definite shape but indefinite volume. b. it has definite shape and definite volume. c. it has indefinite shape and indefinite volume. d. it has indefinite shape but definite volume.
Answers: 1

Chemistry, 23.06.2019 06:20
Why is it that 85.48 rounded to two significant figures is 85 and not 86?
Answers: 1

Chemistry, 23.06.2019 10:20
An engineer wishes to design a container that will hold 12.0 mol of ethane at a pressure no greater than 5.00x10*2 kpa and a temperature of 52.0 degrees celsius. what is the minimum volume the container can have?
Answers: 1
You know the right answer?
A 1 L container originally holds 0.4 mol of N2, 0.1 mol of O2, and 0.08 mole of NO. If the volume of...
Questions

Mathematics, 26.03.2020 22:22

English, 26.03.2020 22:22






Mathematics, 26.03.2020 22:22

Social Studies, 26.03.2020 22:22



Social Studies, 26.03.2020 22:22


Advanced Placement (AP), 26.03.2020 22:22





Mathematics, 26.03.2020 22:23