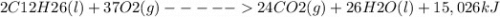Chemistry, 06.08.2021 14:00 leylaanddade
Consider the following thermochemical reaction for kerosene:
2 C12H26(l) + 37 O2(g) 24 CO2(g) + 26 H2O(l) + 15,026 kJ
(a) When 21.3 g of CO2 are made, how much heat is released?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 06:00
What type of electromagnetic radiation has a shorter wavelength than blue light
Answers: 2

Chemistry, 22.06.2019 18:30
The number of moles of a given mass of a substance can be found without knowing its molecular formula or molar mass. true false
Answers: 1

Chemistry, 22.06.2019 21:00
Once similarity and one difference between a mixture of elements and a mixture of compounds
Answers: 3
You know the right answer?
Consider the following thermochemical reaction for kerosene:
2 C12H26(l) + 37 O2(g) 24 CO2(g) + 2...
Questions

Mathematics, 02.08.2019 20:00



History, 02.08.2019 20:00




Health, 02.08.2019 20:00



Physics, 02.08.2019 20:00








Chemistry, 02.08.2019 20:00