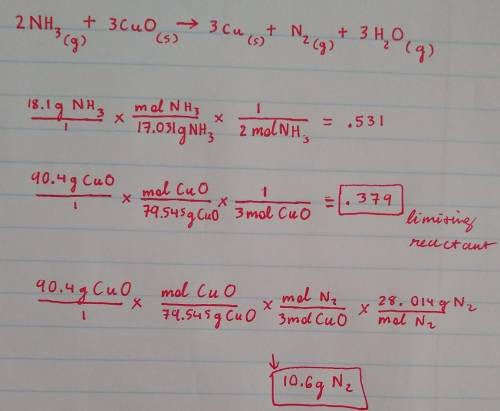Chemistry, 07.08.2021 14:00 zanedog2018
Nitrogen gas can be prepared by passing gaseous ammonia over solid copper (II) oxide at high temperatures. If 18.1 g of Nh3 is reacted with 90.4 g of CuO, which is the limiting reactant? How many grams of N2 will be formed? Explain how you solved for your answers. Balanced Equation: 2NH3 + 3CuO → 3Cu + N2 + 3H2O

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:50
Significant figures are digits read directly from the measuring instrument plus one more digit, which is __ by the observer.
Answers: 2

Chemistry, 22.06.2019 12:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1

Chemistry, 22.06.2019 21:30
Harry lives in a city, and he has a lung condition known as asthma. on certain days, harry has to stay inside because pollutants in the air make it difficult for him to breathe. which of these pollution sources are nonpoint sources that might bother harry if he goes outside? choose the two that apply.
Answers: 3
You know the right answer?
Nitrogen gas can be prepared by passing gaseous ammonia over solid copper (II) oxide at high tempera...
Questions

History, 10.04.2020 22:53




Mathematics, 10.04.2020 22:53



Mathematics, 10.04.2020 22:53

History, 10.04.2020 22:53


Mathematics, 10.04.2020 22:53

Mathematics, 10.04.2020 22:53


Biology, 10.04.2020 22:53


Biology, 10.04.2020 22:53

Mathematics, 10.04.2020 22:53



Mathematics, 10.04.2020 22:54