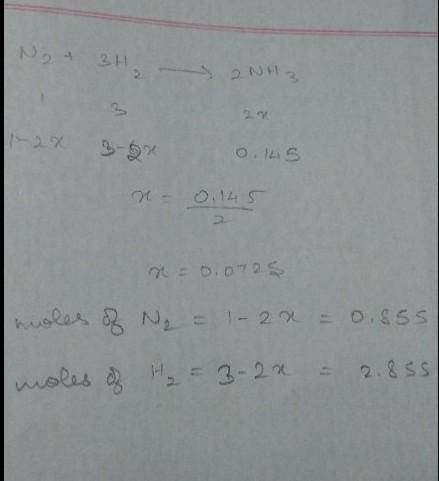Chemistry, 11.08.2021 18:20 maevemboucher78
How many moles of H2 and N2 can be formed by the decomposition of 0.145 mol of ammonia, NH3 ?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:30
When svante arrhenius first proposed his acid-base theory, he was a doctoral candidate. his professors thought his ideas were unfounded. within a decade, the arrhenius theory of acid-base was widely accepted and praised within the scientific world. arrhenius defined acids as compounds having ionizable hydrogen and bases as compounds with ionizable a) barium. b) hydronium. c) hydroxide. d) oxygen.
Answers: 3

Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 11:00
3) in peaches, [oh]=3.16x10-11 m a) find [h+ ] b) what is the ph? c) is the solution acidic, basic, or neutral?
Answers: 1

Chemistry, 22.06.2019 11:20
Which of the following contributes to the structural rigidity of cellulose? adjacent glucose polymers are stabilized by hydrogen bonding. glucose residues are joined by (α1→4) linkages. cellulose is a highly branched molecule. the conformation of the glucose polymer is a coiled structure.
Answers: 2
You know the right answer?
How many moles of H2 and N2 can be formed by the decomposition of 0.145 mol of ammonia, NH3 ?...
Questions

Mathematics, 08.01.2020 20:31



Mathematics, 08.01.2020 20:31


History, 08.01.2020 20:31



English, 08.01.2020 20:31

Mathematics, 08.01.2020 20:31

Biology, 08.01.2020 20:31

Mathematics, 08.01.2020 20:31


Business, 08.01.2020 20:31

Business, 08.01.2020 20:31

Computers and Technology, 08.01.2020 20:31

Biology, 08.01.2020 20:31


English, 08.01.2020 21:31