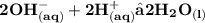Chemistry, 18.08.2021 14:00 allisonboggs85
A student titrated a solution containing 3.7066 g of an unknown Diprotic acid to the end point using 28.94 ml of 0.3021 M KOH solution. What is the molar mass of the unknown acid? Hint: you must write a balanced equation for the reaction.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:30
What royal scientist used the 29th day of frozen vapor to encounter elements for mastering new culinary creations?
Answers: 1

Chemistry, 22.06.2019 05:40
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 17:00
Which property of a rock remains unchanged by mechanical weathering? a. total surface area b. size and shape c. mineral composition d. sharpness
Answers: 1

Chemistry, 22.06.2019 19:10
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2
You know the right answer?
A student titrated a solution containing 3.7066 g of an unknown Diprotic acid to the end point using...
Questions

English, 20.12.2019 11:31

Biology, 20.12.2019 11:31



Mathematics, 20.12.2019 11:31

History, 20.12.2019 11:31


Physics, 20.12.2019 11:31


Mathematics, 20.12.2019 11:31






Mathematics, 20.12.2019 11:31

Mathematics, 20.12.2019 11:31

Mathematics, 20.12.2019 11:31