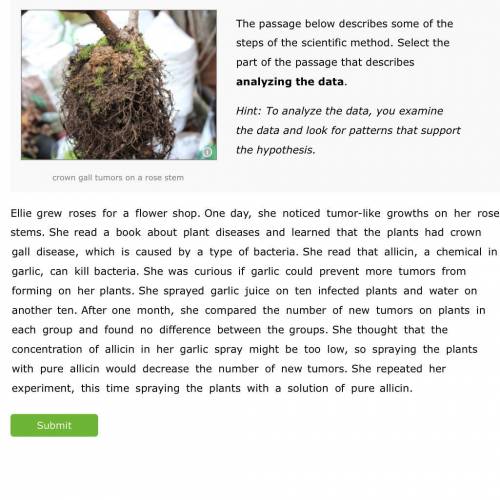
Can y’all help me on this
...

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2


Chemistry, 23.06.2019 01:00
Heat energy, carbon dioxide, and water are released through which process? a. photosynthesis b. depolymerization c. digestion d. cellular respiration
Answers: 1

Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1
You know the right answer?
Questions


World Languages, 05.07.2019 19:30



Mathematics, 05.07.2019 19:30

English, 05.07.2019 19:30


Mathematics, 05.07.2019 19:30


Computers and Technology, 05.07.2019 19:30


Mathematics, 05.07.2019 19:30

Mathematics, 05.07.2019 19:30

Computers and Technology, 05.07.2019 19:30



English, 05.07.2019 19:30

Mathematics, 05.07.2019 19:30

Mathematics, 05.07.2019 19:30