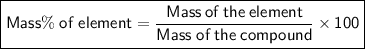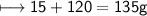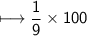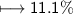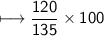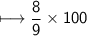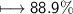Chemistry, 01.09.2021 14:00 Zagorodniypolina5
compound 1 contains 15.0g of hydrogen and 120.0g oxygen. What is the percent compound of each element?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 04:00
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 07:00
At 450 mm hg a gas has a volume of 760 l, what is its volume at standard pressure
Answers: 2

Chemistry, 22.06.2019 10:50
A100 kmol/h stream that is 97 mole% carbon tetrachloride (ccl4) and 3% carbon disulfide (cs2) is to be recovered from the bottom of a distillation column. the feed to the column is 16 mole% cs2 and 84% ccl4, and 2% of the ccl4 entering the column is contained in the overhead stream leaving the top of the column. calculate the mass and mole fractions of ccl4 in the overhead stream, and determine the molar flow rates of ccl4 and cs2 in the overhead and feed streams. 12. mw_ccla- 153.82; mw_cs2-76.14.
Answers: 3

Chemistry, 23.06.2019 00:00
How many moles of water are created if 3 moles of hydrogen react completely with excess oxygen?
Answers: 3
You know the right answer?
compound 1 contains 15.0g of hydrogen and 120.0g oxygen. What is the percent compound of each elemen...
Questions

Social Studies, 19.10.2019 00:20

Mathematics, 19.10.2019 00:20



Mathematics, 19.10.2019 00:20



History, 19.10.2019 00:20

Mathematics, 19.10.2019 00:20


English, 19.10.2019 00:20




Mathematics, 19.10.2019 00:20


English, 19.10.2019 00:20


English, 19.10.2019 00:20