
Chemistry, 06.09.2021 05:50 destiny465
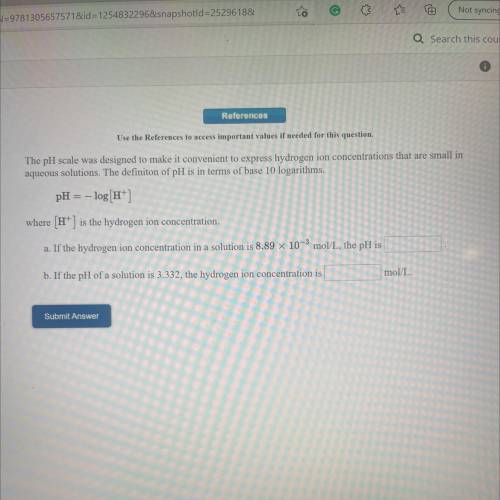
Use the References to access important values if needed for this question.
H
A-Z
The pH scale was designed to make it convenient to express hydrogen ion concentrations that are small in
aqueous solutions. The definiton of pH is in terms of base 10 logarithms.
pH = -log[H+]
where (H+) is the hydrogen ion concentration.
a. If the hydrogen ion concentration in a solution is 8.89 x 10-3 mol/L, the pH is
b. If the pH of a solution is 3.332, the hydrogen ion concentration is
mol/L.
Submit Answer

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:10
Harvey mixes two liquids. which observation of the new mixture most likely indicates a precipitate is forming?
Answers: 2



Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1
You know the right answer?
Use the References to access important values if needed for this question.
H
A-Z
The p...
A-Z
The p...
Questions

Mathematics, 26.04.2021 06:40


Chemistry, 26.04.2021 06:40



Mathematics, 26.04.2021 06:40





Mathematics, 26.04.2021 06:40

Mathematics, 26.04.2021 06:40





Mathematics, 26.04.2021 06:40


Business, 26.04.2021 06:40

Mathematics, 26.04.2021 06:40



