Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:30
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible?
Answers: 2

Chemistry, 21.06.2019 23:00
Will mark brainliest26. which of these statements are true? (3 points)a. gases are compressibleb. gases fill their containers completelyc. the pressure of a gas is independent of the temperatured. gases have masse. gases exert pressuref. the pressure of a gas is dependent on the volumeg. gas pressure results from the collisions between gas particlesh. gases have a definite volume and shape
Answers: 1

Chemistry, 22.06.2019 09:20
Explain that newton first law,second law and third law of motion?
Answers: 2

Chemistry, 22.06.2019 22:30
Rank the four gases (air, exhaled air, gas produced from from decomposition of h2o2, gas from decomposition of nahco3) in order of increasing concentration of co2
Answers: 1
You know the right answer?
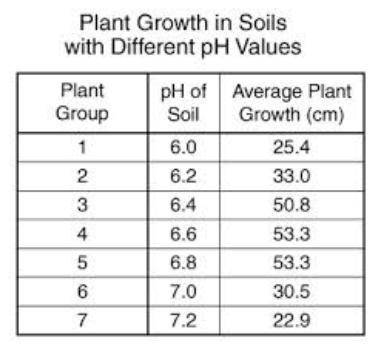
30. What was the range in average plant growth?
31. What is the average value of the average plant...
Questions



Mathematics, 01.01.2020 23:31


History, 01.01.2020 23:31


History, 01.01.2020 23:31


History, 01.01.2020 23:31



History, 01.01.2020 23:31

Biology, 01.01.2020 23:31

History, 01.01.2020 23:31

Social Studies, 01.01.2020 23:31

Mathematics, 01.01.2020 23:31


History, 01.01.2020 23:31

History, 01.01.2020 23:31

History, 01.01.2020 23:31