
Chemistry, 07.09.2021 23:40 wwwclarajohnson39
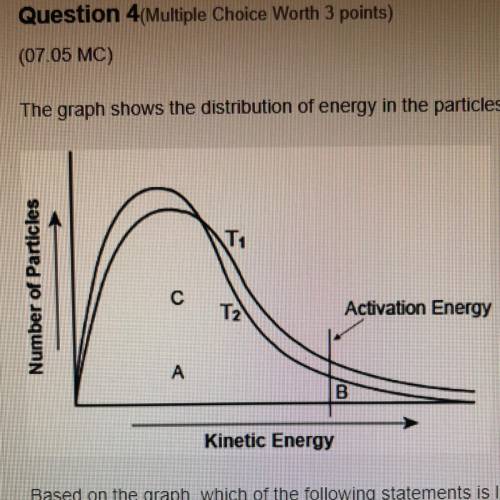
The graph shows the distribution of energy in the particles of two gas samples at different temperatures, T and T2. A, B, and C represent individual particles
Based on the graph, which of the following statements is likely to be true?
Particle A and C are more likely to participate in the reaction than particle B.
Most of the particles of the two gases have very high speeds.
A fewer number of particles of gas at T, are likely to participate in the reaction than the gas at T2
The average speed of gas particles at T2 is lower than the average speed of gas particles at T1

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 16:00
About 3% of the water on earth is freshest. only about 40% of that freshwater is available for human use. why is so much freshwater unavailable for human use?
Answers: 2

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 22.06.2019 20:30
Which of the following is not true about the atomic model of substances?
Answers: 1

Chemistry, 22.06.2019 22:30
Draw the aromatic compound toluene (methylbenzene). show all hydrogen atoms, including those on the ring.
Answers: 1
You know the right answer?
The graph shows the distribution of energy in the particles of two gas samples at different temperat...
Questions

History, 17.10.2020 23:01


Mathematics, 17.10.2020 23:01


History, 17.10.2020 23:01

Business, 17.10.2020 23:01


Mathematics, 17.10.2020 23:01

Biology, 17.10.2020 23:01

Geography, 17.10.2020 23:01

Mathematics, 17.10.2020 23:01





Mathematics, 17.10.2020 23:01

Biology, 17.10.2020 23:01

Physics, 17.10.2020 23:01


English, 17.10.2020 23:01



