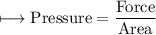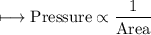Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 22.06.2019 21:00
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1

Chemistry, 23.06.2019 03:40
The following questions a24 - a26 relate to 100 ml of 0.0150 m solution of benzoic acid (c6h3cooh). ka(c6h3cooh) = 6.4 x 10^-5. what is the ph of the solution after the addition of 1 x 10^-3 moles of naoh? you may assume no volume change to the solution upon addition of the naoh.
Answers: 2
You know the right answer?
Illustrate the increase in rate of reaction with the increase in surface area
more examples in dai...
Questions

Mathematics, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01

Biology, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01

Social Studies, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01

English, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01

Physics, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01

Mathematics, 11.09.2020 17:01