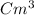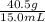Chemistry, 04.10.2021 20:30 Victoriag2626
50 Points Help
1) A block of aluminum occupies a volume of 15.0 mL and mass 40.5 g. What is its density?
2) Mercury metal is poured into a graduated cylinder that holds exactly 22.5 mL. The mercury used to fill the cylinder mass 306.0 g. From this information, calculate the density of mercury.
3) What is the mass of the ethyl alcohol that exactly fills a 200.0 mL container? The density of ethyl alcohol is 0.789 g/mL.
4) A rectangular block of copper metal weighs 1896 g. The dimensions of the block are 8.4 cm by 5.5 cm by 4.6 cm. From this data, what is the density of copper? ( hint find the volume first: volume of rectangle = Hight x width x length)
5) Calculate the density of sulfuric acid if 35.4 mL of the acid mass 65.14 g.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 1


Chemistry, 22.06.2019 23:00
In the reaction h2co3 (aq) + 3nh3 (aq) = 2 nh4+ (aq) + co3 2-, how many electrons are transferred?
Answers: 3

Chemistry, 22.06.2019 23:10
Afusion reaction takes place between carbon and another element. neutrons are released, and a different element is formed. the different element is a) lighter than helium.b)heavier than helium.c)the same weight as helium.d)dependent on the element that reacted with carbon.
Answers: 3
You know the right answer?
50 Points Help
1) A block of aluminum occupies a volume of 15.0 mL and mass 40.5 g. What is its de...
Questions

SAT, 04.01.2022 17:30

Mathematics, 04.01.2022 17:30


Geography, 04.01.2022 17:30

Chemistry, 04.01.2022 17:30



History, 04.01.2022 17:30

English, 04.01.2022 17:30


Mathematics, 04.01.2022 17:30

Mathematics, 04.01.2022 17:30



Mathematics, 04.01.2022 17:30

SAT, 04.01.2022 17:30

Advanced Placement (AP), 04.01.2022 17:30

SAT, 04.01.2022 17:30


English, 04.01.2022 17:30

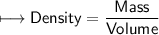

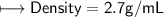

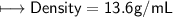

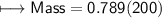
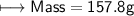
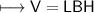
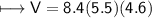
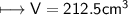
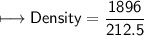
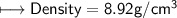
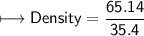
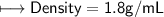
 when dealing with a solid.
when dealing with a solid.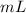 or
or