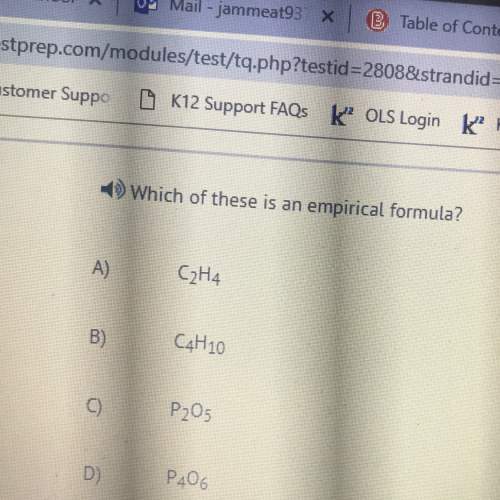
Chemistry, 08.10.2021 22:50 smortandsons
A student dissolves 2.40g of impure sulfamic acid in water and makes the solution up to 250cm3. He titrates the sulfamic acid solution with 25.0cm3 aliquots of 0.0700mol dm–3 sodium hydroxide solution. The mean titre is 17.95cm3. (a) Calculate the percentage purity of the sulfamic acid.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:30
The table lists pressure and volume values for a particular gas. which is the best estimate for the value of v at p = 7.0 × 103 pascals?
Answers: 3


Chemistry, 22.06.2019 10:10
Stage in which a star’s outer layers have started to cool and grow outward?
Answers: 3

Chemistry, 22.06.2019 20:00
What is the molarity of the solution produced when 145 g of nacl is dissolved in sufficient water to prepare 2.75 l of solution?
Answers: 1
You know the right answer?
A student dissolves 2.40g of impure sulfamic acid in water and makes the solution up to 250cm3. He t...
Questions


Mathematics, 26.10.2020 01:40

Mathematics, 26.10.2020 01:40



History, 26.10.2020 01:40



Biology, 26.10.2020 01:40


Mathematics, 26.10.2020 01:40

Engineering, 26.10.2020 01:40


Physics, 26.10.2020 01:40

Mathematics, 26.10.2020 01:40

Chemistry, 26.10.2020 01:40

Chemistry, 26.10.2020 01:40


Social Studies, 26.10.2020 01:40