
Chemistry, 09.10.2021 14:00 magmoo3779
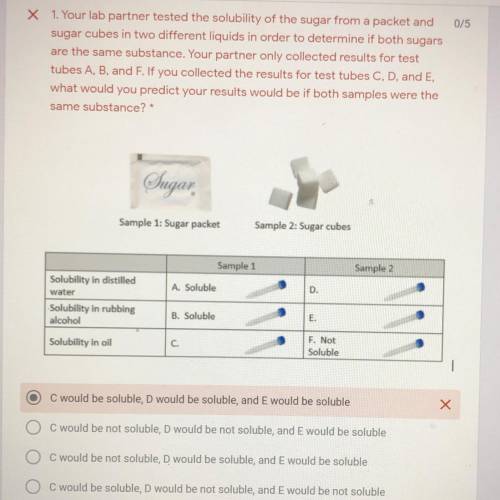
Your lab partner tested the solubility of a sugar from a packet and sugar cubes in two different liquids in order to determine if both sugars are the same substance. Your partner only collected the results for the test tubes A, B, and F. If you collected the results for C D and E, what would you predict your results would be if both samples were the same substance
A. C would be soluble, D would be soluble, and E would be soluble
B. C would be not soluble, D would be not soluble, and E would be soluble
C. C would be not soluble, D would be soluble, and E would be soluble.
D. C would be soluble, D would not be soluble, and E would be not soluble

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1

Chemistry, 22.06.2019 13:50
How does the motion of particles in a gas change as the gas cools
Answers: 2


Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1
You know the right answer?
Your lab partner tested the solubility of a sugar from a packet and sugar cubes in two different liq...
Questions

Mathematics, 01.04.2021 03:30



Arts, 01.04.2021 03:30


English, 01.04.2021 03:30

Mathematics, 01.04.2021 03:30

Mathematics, 01.04.2021 03:30


Spanish, 01.04.2021 03:30


Biology, 01.04.2021 03:30

History, 01.04.2021 03:30

Mathematics, 01.04.2021 03:30


History, 01.04.2021 03:30






