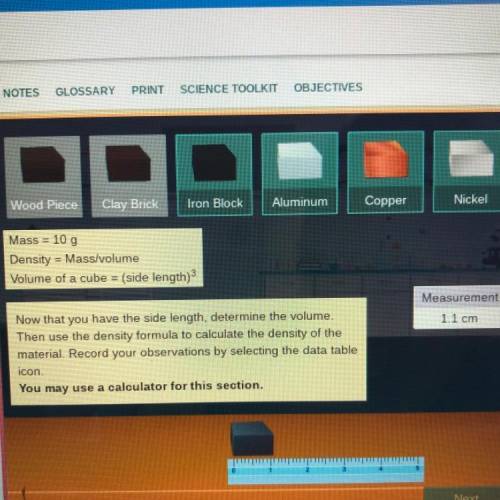
Mass = 10 g
Density = Mass/volume
Volume of a cube = (side length)3
Measurement
...

Chemistry, 09.10.2021 21:40 jaychavez1926
Mass = 10 g
Density = Mass/volume
Volume of a cube = (side length)3
Measurement
1.1 cm
Now that you have the side length, determine the volume.
Then use the density formula to calculate the density of the
material. Record your observations by selecting the data table
icon.
You may use a calculator for this section.
Measurement
1.1 cm
Please help me explain to me how you can determine the Volume I don’t know how to do that and I need help ASAP today is the last day to do this and I really need help

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 16:40
Which of the following is a testable hypothesis? a. if i brush my teeth, i will get fewer cavities than if i don't brush my teeth. b. green toothpaste tastes better than blue toothpaste or red toothpaste. c. smart, careful, healthy people always brush their teeth. d. it's wrong to not brush your teeth before you have an important conversation with someone.
Answers: 1

Chemistry, 22.06.2019 02:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al
Answers: 1

Chemistry, 22.06.2019 05:30
What happens to the atomic radius when an elctron is lost
Answers: 1

Chemistry, 22.06.2019 20:00
There are two steps in the usual industrial preparation of acrylic acid, the immediate precursor of several useful plastics. in the first step, calcium carbide and water react to form acetylene and calcium hydroxide: cac2 (s) + 2h2o (g) → c2h2 (g) + caoh2 (s) =δh−414.kj in the second step, acetylene, carbon dioxide and water react to form acrylic acid: 6c2h2 (g) + 3co2 (g) + 4h2o (g) → 5ch2chco2h (g) =δh132.kj calculate the net change in enthalpy for the formation of one mole of acrylic acid from calcium carbide, water and carbon dioxide from these reactions. round your answer to the nearest kj .
Answers: 3
You know the right answer?
Questions





Mathematics, 16.12.2019 20:31

Spanish, 16.12.2019 20:31

Mathematics, 16.12.2019 20:31




Mathematics, 16.12.2019 20:31




Spanish, 16.12.2019 20:31







