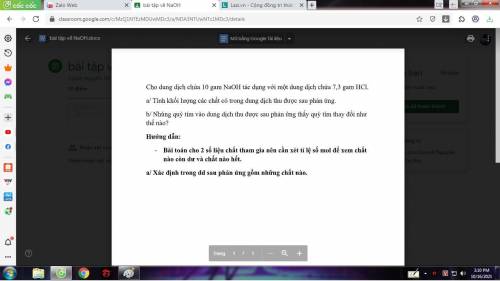
Giúp mình có thể giải thích
...

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Construct the hypothetical phase diagram for metals a and b between room temperature (20c) and 700c, given the following information: * the melting temperature of metal a is 480c. • the maximum solubility of b in a is 4 wt% b, which occurs at 420c. • the solubility of b in a at room temperature is 0 wt% b. • one eutectic occurs at 420c and 18 wt% b–82 wt% a. • a second eutectic occurs at 475c and 42 wt% b–58 wt% a. • the intermetallic compound ab exists at a composition of 30 wt% b–70 wt% a, and melts congruently at 525c.• the melting temperature of metal b is 600c. • the maximum solubility of a in b is 13 wt% a, which occurs at 475c. • the solubility of a in b at room temperature is 3 wt% a.
Answers: 1


Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 08:30
Which change in temperature is the smallest? a change of 1 thomson degree a change of 1 kelvin degree a change of 1 fahrenheit degree a change of 1 celsius degree
Answers: 1
You know the right answer?
Questions

Chemistry, 05.03.2021 14:00

English, 05.03.2021 14:00

Physics, 05.03.2021 14:00



Mathematics, 05.03.2021 14:00





Mathematics, 05.03.2021 14:00

Mathematics, 05.03.2021 14:00

Geography, 05.03.2021 14:00


Biology, 05.03.2021 14:00

Computers and Technology, 05.03.2021 14:00


Mathematics, 05.03.2021 14:00

Physics, 05.03.2021 14:00

Mathematics, 05.03.2021 14:00