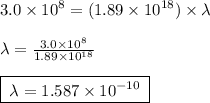Chemistry, 27.10.2021 18:10 avengegaming7
Please help me I can’t figure it out
1. Calculate the wavelength of light with a frequency of 1.89 x 1018 Hz.
a

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:30
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1


Chemistry, 23.06.2019 05:30
The image compares the arrangement of electrons in two different neutral atoms. a figure labeled atom q has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has six black spheres. to the left of this figure is another figure labeled atom p. atom p has a shaded sphere at the center of three concentric circles. the innermost circle has two black spheres. the middle circle has seven black spheres. which of the following best explains the position of the two atoms in the periodic table? atom p has an estimated zeff of 7 and is therefore to the left of atom q, which has a zeff of 6. atom p has an estimated zeff of 7 and is therefore to the right of atom q, which has a zeff of 6. atom p has an estimated zeff of 5 and is therefore below atom q, which has a zeff of 4. atom p has an estimated zeff of 5 and is therefore above atom q, which has a zeff of 4.
Answers: 3

Chemistry, 23.06.2019 15:30
Acontainer holds 6.4 moles of gas. hydrogen gas makes up 25% of the total moles in the container. if the total pressure is 1.24atm. what is the partial pressure of hydrogen
Answers: 3
You know the right answer?
Please help me I can’t figure it out
1. Calculate the wavelength of light with a frequency of 1.89...
Questions




Physics, 02.09.2019 08:30

Chemistry, 02.09.2019 08:30

Social Studies, 02.09.2019 08:30

Mathematics, 02.09.2019 08:30



Mathematics, 02.09.2019 08:30

Physics, 02.09.2019 08:30


Mathematics, 02.09.2019 08:30