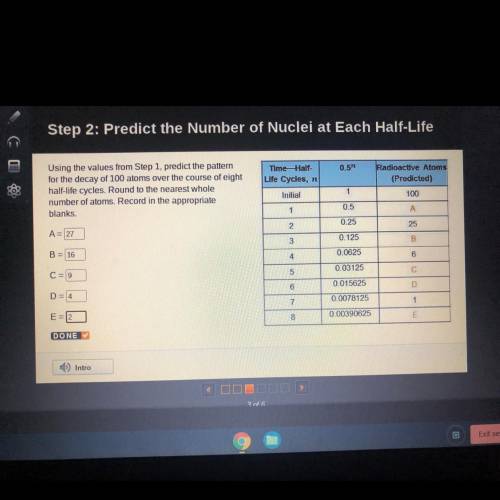
Step 2: Predict the Number of Nuclei at Each Half-Life
0.5"
Using the values from Step 1, pr...

Chemistry, 29.11.2021 05:20 vannaht2003
Step 2: Predict the Number of Nuclei at Each Half-Life
0.5"
Using the values from Step 1, predict the pattern
for the decay of 100 atoms over the course of eight
half-life cycles, Round to the nearest whole
number of atoms. Record in the appropriate
blanks
Time- Half-
Life Cycles, no
Initial
Radioactive Atoms
(Predicted)
100
1
1
0.5
А
2
0.25
A= 27
25
3
0.125
B
B = 16
4.
0.0625
6
C = 9
5
0.03125
С
D=4
6
0.015625
D
7
0.0078125
1
E
8
0.00390625
E
DONE
) Intro

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 02:30
Which compound contains both ionic and covalent bonds? a) hbr b)cbr4 c)nabr d) naoh
Answers: 2

Chemistry, 22.06.2019 04:40
Listen base your answer to the question on the information below.propane is a fuel that is sold in rigid, pressurized cylinders. most of the propane in a cylinder is liquid, with gas in the space above the liquid level. when propane is released from the cylinder, the propane leaves the cylinder as a gas. propane gas is used as a fuel by mixing it with oxygen in the air and igniting the mixture, as represented by the balanced equation below.c3h8(g) + 5o2(g) → 3co2(g) + 4h2o() + 2219.2 kja small amount of methanethiol, which has a distinct odor, is added to the propane to consumers detect a propane leak. in methanethiol, the odor is caused by the thiol functional group (–sh). methanethiol, ch3sh, has a structure that is very similar to the structure of methanol.what is the correct structural formula for a molecule of methanethiol
Answers: 3

Chemistry, 22.06.2019 11:30
If blood contains 150g of hemoglobin per liter of blood, how much hemoglobin would be contained in 10 ml of blood
Answers: 2
You know the right answer?
Questions








Mathematics, 03.03.2020 05:42

Mathematics, 03.03.2020 05:42













