
Chemistry, 10.12.2021 04:10 mylittleponeyfbrarit
Prediction: The Cucl, crystal will dissolve in water. If we write the solvation process as a chemical
equation, we can predict two possible outcomes:
a. Cuci
H2O Cu?* (aq) + 2 Cl(aq)
H2O Cu2+ (aq) + Cl2(9)
+ 9
.
b. Cuciz
In equation (a) Cl' ions remain dissolved in water whereas in equation (b) the chlorine molecule CI,
forms and leaves the solution as a gas. Which outcome do you expect (a or b)? Explain your reasoning.
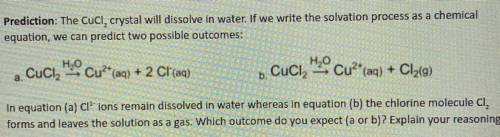

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

Chemistry, 23.06.2019 06:50
What is the volume of 3.2 moles of chlorine gas (cl2) at 295 k and 1.1 atm?
Answers: 1


Chemistry, 23.06.2019 10:10
In a covalent bond, two atoms are held together by the attraction between . the number of covalent bonds that an atom can form depends on the number of in the atom.
Answers: 2
You know the right answer?
Prediction: The Cucl, crystal will dissolve in water. If we write the solvation process as a chemica...
Questions



Mathematics, 25.08.2021 06:00

Geography, 25.08.2021 06:00

Mathematics, 25.08.2021 06:00



Mathematics, 25.08.2021 06:00

Mathematics, 25.08.2021 06:00

Mathematics, 25.08.2021 06:00


English, 25.08.2021 06:00


Mathematics, 25.08.2021 06:00

Chemistry, 25.08.2021 06:00


Mathematics, 25.08.2021 06:00

Mathematics, 25.08.2021 06:00




