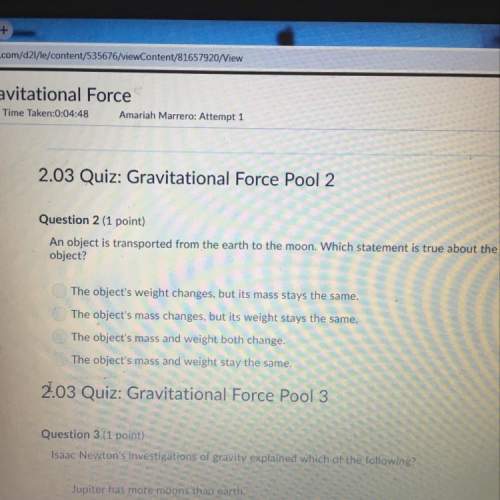
Chemistry, 10.12.2021 22:00 sharpeyennifer
Calculate the pressure in kPa exerted by 0.300 mol of acetic acid in a 2 L container at 40OC
a) Using ideal gas law behavior
b) Van der Waals equation, given a = 17.71 atm. L2/mol2 and b = 0.0237L. mol.
c) Which pressure is the most accurate and why?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:00
The compound methyl butanoate smells like apples. its percent composition is 58.8% c, 9.9% h, and 31.4% o. what’s the empirical formula ?
Answers: 1

Chemistry, 22.06.2019 05:00
Type the letter that represents the correct location for each particle type below.
Answers: 1

Chemistry, 22.06.2019 20:30
The activation energy for the reaction no2(g)+co2(g)⟶no(g)+co(g) is ea = 300 kj/mol and the change in enthalpy for the reaction is δh = -100 kj/mol . what is the activation energy for the reverse reaction?
Answers: 3

Chemistry, 22.06.2019 21:00
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1
You know the right answer?
Calculate the pressure in kPa exerted by 0.300 mol of acetic acid in a 2 L container at 40OC
a) Us...
Questions

Mathematics, 18.10.2019 23:30


History, 18.10.2019 23:30



English, 18.10.2019 23:30


Advanced Placement (AP), 18.10.2019 23:30



Mathematics, 18.10.2019 23:30