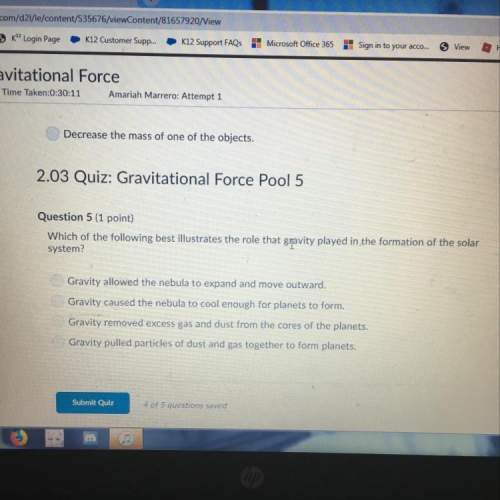
Chemistry, 13.12.2021 18:00 tristanortonubca
How do chemists predict the shapes of molecules?
Chemists can't predict the shape of molecules, because the attractive forces between valence electrons are unpredictable.
Due to the repulsion between electrons, valence electrons will be arranged as close to each other as possible.
Due to the attraction between electrons, valence electrons will be arranged as close to each other as possible.
Due to the repulsion between electrons, valence electrons will be arranged as far apart from each other as possible.

Answers: 2


Another question on Chemistry



Chemistry, 22.06.2019 14:30
Connect the whole numbers on the periodic table to indicate what they represent?
Answers: 3

Chemistry, 22.06.2019 23:00
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm?
Answers: 3
You know the right answer?
How do chemists predict the shapes of molecules?
Chemists can't predict the shape of molecules, be...
Questions



English, 12.09.2019 08:20

Mathematics, 12.09.2019 08:20





Mathematics, 12.09.2019 08:20


Mathematics, 12.09.2019 09:10

Mathematics, 12.09.2019 09:10



Biology, 12.09.2019 09:10

Mathematics, 12.09.2019 09:10




Mathematics, 12.09.2019 09:10