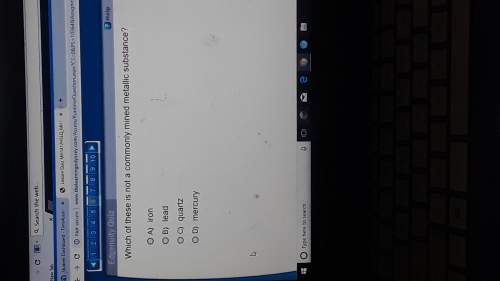
Chemistry, 19.12.2021 16:20 michaelgibsonsto
Zn + 2HCl —> ZnCl2 + H2 If you have 6.8 moles of zinc chloride produced, how many moles of zinc reacted?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 12:00
Under normal conditions, describe how increasing the temperatures effects the solubility of a typical salt
Answers: 1

Chemistry, 22.06.2019 16:10
Given the following equation: 2a1 + 3mgcl2 --> 2alcl3 + 3mg how many moles of aluminum chloride are produced from 2.5 moles of magnesium chloride?
Answers: 1

Chemistry, 22.06.2019 18:00
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2
You know the right answer?
Zn + 2HCl —> ZnCl2 + H2
If you have 6.8 moles of zinc chloride produced, how many moles of zinc...
Questions



History, 07.04.2021 01:00

Mathematics, 07.04.2021 01:00




Mathematics, 07.04.2021 01:00

Mathematics, 07.04.2021 01:00



Mathematics, 07.04.2021 01:00


Mathematics, 07.04.2021 01:00

Mathematics, 07.04.2021 01:00


Mathematics, 07.04.2021 01:00

Mathematics, 07.04.2021 01:00


Mathematics, 07.04.2021 01:00