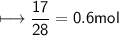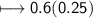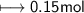Chemistry, 16.01.2022 05:50 ashleyvalles16
Calculate the number of moles of SO2 required to produce 17 g of CO in the following chemical equation: 5 C + 2 SO2 → CS2 + 4 CO

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 3

Chemistry, 22.06.2019 08:40
Which statement can best be concluded from the ideal gas law?
Answers: 2


Chemistry, 22.06.2019 20:30
Some familiar products contain some of the same types of atoms. for instance, the chemical formula for baking soda is nahco 3. the chemical formula for liquid bleach is naclo, and the chemical formula for table salt is nacl. which choice best describes why these three products have some of the same types of atoms in common?
Answers: 1
You know the right answer?
Calculate the number of moles of SO2 required to produce 17 g of CO in the following chemical equati...
Questions


Health, 24.07.2019 10:30

Mathematics, 24.07.2019 10:30


History, 24.07.2019 10:30









Mathematics, 24.07.2019 10:30

Mathematics, 24.07.2019 10:30