
Chemistry, 13.02.2022 04:40 DesireeGuzman5213
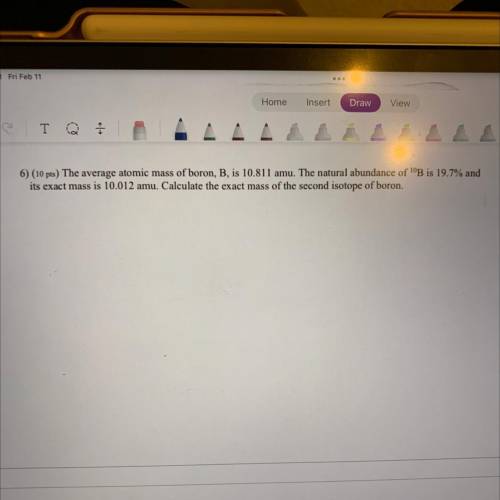
6) The average atomic mass of boron, B, is 10.811 amu. The natural abundance of 10B is 19.7% and it’s exact mass is 10.012 amu. Calculate the exact mass of the second isotope of boron.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:50
How many liters of oxygen gas, at standard temperature and pressure, will react with 35.8 grams of iron metal? 4 fe (s) + 3 o₂ (g) → 2 fe₂o₃ (s)
Answers: 2


Chemistry, 22.06.2019 14:30
How do temperature and salinity affect deepwater currents? as temperatures and salinity levels of water increase, the water rises to the surface where it creates currents as it moves to colder regions. they create changes in wind direction, moving denser water in the same direction as the wind and causing the deepwater circulation patterns found in the ocean. they equalize the forces on undersea currents caused by the coriolis effect as they replace more dense water with less dense water. they create density differences that cause dense deepwater currents to flow toward the equator where they displace less dense, warmer water above them.
Answers: 2

Chemistry, 23.06.2019 01:00
Who examines and coordinates the cleanup of polluted sites?
Answers: 2
You know the right answer?
6) The average atomic mass of boron, B, is 10.811 amu. The natural abundance of 10B is 19.7% and it’...
Questions

History, 12.07.2019 17:00

History, 12.07.2019 17:00

English, 12.07.2019 17:00


Health, 12.07.2019 17:00


Mathematics, 12.07.2019 17:00

Mathematics, 12.07.2019 17:00


Mathematics, 12.07.2019 17:00

History, 12.07.2019 17:00


Health, 12.07.2019 17:00


Social Studies, 12.07.2019 17:00

History, 12.07.2019 17:00

Mathematics, 12.07.2019 17:00

History, 12.07.2019 17:00


Mathematics, 12.07.2019 17:00



