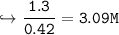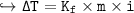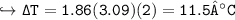5 points
15. Given the formula below: What is the change in the water's freezing
point when...

Chemistry, 22.02.2022 03:20 keilahsalmon
5 points
15. Given the formula below: What is the change in the water's freezing
point when 75 grams of NaCl is added to 400 grams of H20? (Kf water -
1.86 C/m) *
Change in Temp = - ix K, x m
- 0.119 ° C
- 1.19 ° C
O A
B.
- 11.9°C
- 119°C
C

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:20
What is the strongest intermolecular force between an nacl unit and an h2o molecule together in a solution? covalent bonding dipole-dipole force hydrogen bonding ion-dipole force
Answers: 1

Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3

Chemistry, 22.06.2019 16:30
Explain in detail of the four major scientific developments that spurred the formulation of the plate tectonics theory
Answers: 2

Chemistry, 22.06.2019 17:40
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1
You know the right answer?
Questions



Mathematics, 25.09.2019 09:00




Physics, 25.09.2019 09:00

Mathematics, 25.09.2019 09:00


History, 25.09.2019 09:00

Mathematics, 25.09.2019 09:00

Mathematics, 25.09.2019 09:00

History, 25.09.2019 09:00


History, 25.09.2019 09:00

Mathematics, 25.09.2019 09:00


History, 25.09.2019 09:00